These clinical areas reflect where ongoing research highlights the role of neural–immune balance in long-term health.
Rheumatoid Arthritis

What is rheumatoid arthritis?
Rheumatoid arthritis is a chronic autoimmune disease in which the immune system mistakenly attacks its own tissues, primarily the lining of the joints. This causes chronic inflammation that leads to pain, stiffness, swelling, and, over time, progressive joint damage and reduced movement.
Common symptoms
People living with rheumatoid arthritis often experience:
- Persistent joint pain, swelling, and tenderness
- Reduced mobility and weakness
- Fatigue
- Periods of flares followed by partial relief
When inflammation doesn’t switch off
In a healthy body, inflammation turns on to fight injury or infection—and then turns off. In rheumatoid arthritis, this “off switch” does not work properly. The immune system remains chronically active, continuously releasing inflammatory signaling molecules such as cytokines (including TNF). This ongoing inflammation drives pain, fatigue, joint damage, and systemic inflammation involving blood vessels and the heart. Research shows that this process is closely linked to impaired regulation by the nervous system, not only immune dysfunction.
The role of the vagus nerve
The vagus nerve system is a major communication pathway between the brain and the immune system. It plays a central role in controlling inflammation through a built-in mechanism called the inflammatory reflex. In people with rheumatoid arthritis, vagal activity (often referred to as vagal tone) is commonly reduced. This means the body’s natural ability to calm excessive inflammation is weakened—contributing to persistent symptoms and disease activity.
What clinical research shows
Research from Feinstein Institutes for Medical Research has demonstrated that vagus nerve stimulation (VNS) can restore this regulatory pathway and reduce inflammation in rheumatoid arthritis. A large randomized, placebo-controlled clinical trial involving 242 patients with moderate-to-severe RA, who had not responded adequately to established pharmacological therapy showed that invasive VNS with a surgically implanted device was safe and effective. This recently led to FDA approval of the first vagus nerve stimulation device for rheumatoid arthritis—marking the first regulatory recognition of activating the inflammatory reflex as a treatment strategy.
Emerging evidence for non-invasive ear-based VNS
Smaller clinical studies indicate that non-invasive stimulation of the cymba conchae, a specific area of the outer ear exclusively innervated by vagus nerve fibers, may also benefit patients with uncontrolled RA. In pilot studies, non-invasive cymba conchae stimulation was:
- Well tolerated
- Associated with meaningful improvements in disease activity
- Linked to reductions in inflammatory markers
- Accompanied by improvements in pain and joint stiffness
Placebo-controlled trials are currently ongoing to further evaluate this approach. Cymbathera AB sponsors one such ongoing randomized, sham-controlled RA study.
Key takeaways
- Rheumatoid arthritis involves both immune dysfunction and impaired nervous system regulation
- Reduced vagus nerve activity contributes to chronic inflammation and pain
- Clinical evidence shows that activating the vagus nerve can reduce inflammation and improve symptoms
- Non-invasive ear-based vagus nerve stimulation is an active area of clinical research
Studies with cymba conchae-mediated VNS
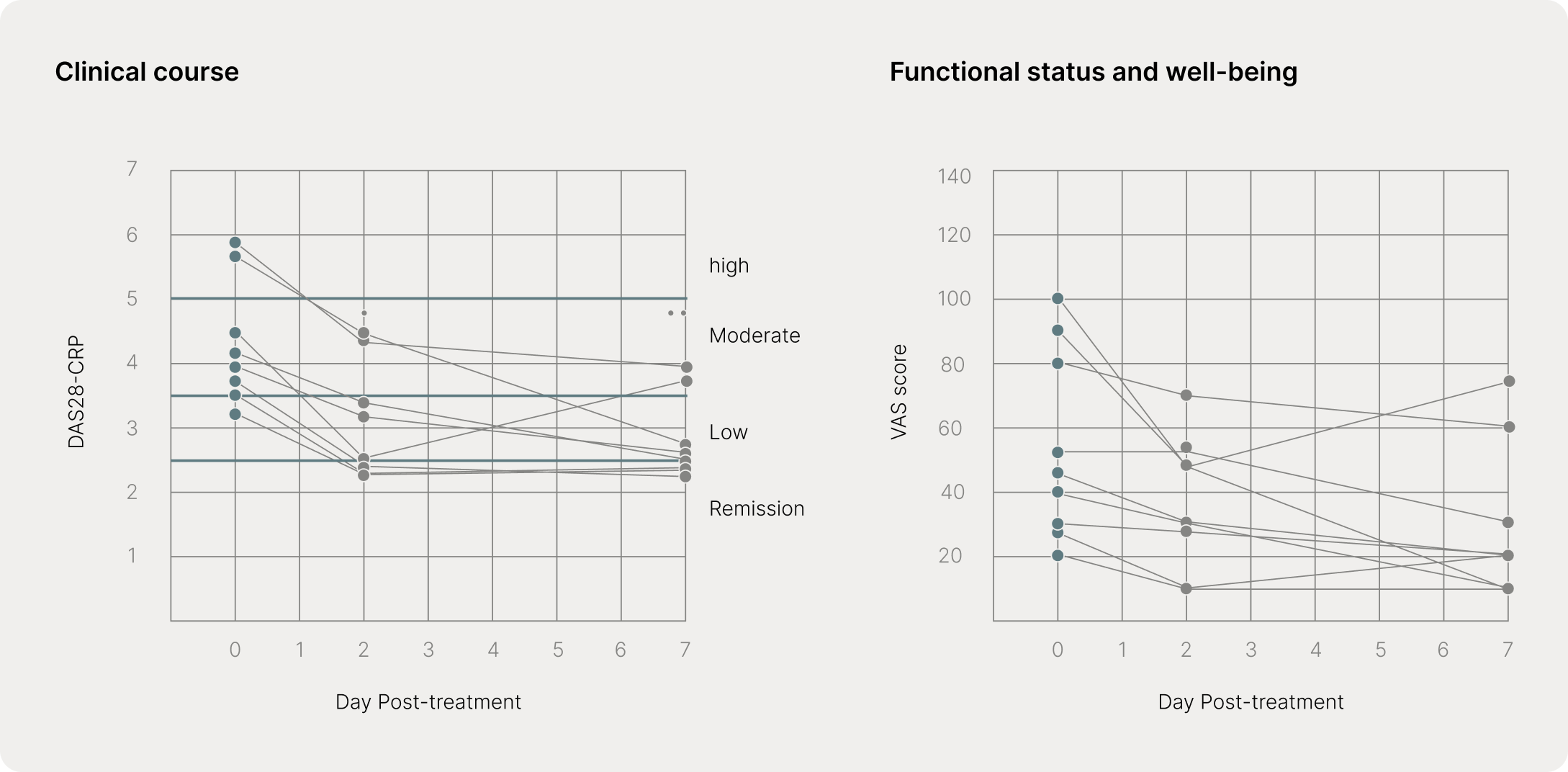
In a pilot study of nine patients with active rheumatoid arthritis, short-term vibrotactile stimulation of the auricular cymba conchae reduced inflammatory disease activity (DAS28-CRP) and improved patient-reported well-being (VAS). These findings demonstrated rapid engagement of vagus nerve–mediated anti-inflammatory pathways following targeted ear stimulation.
Fig. The effect of vibrotactile treatment in the auricular cymba concha area in RA patients. The DAS28-CRP scores indicate a composite score of enumeration of swollen and tender joints, and measurement of high-sensitivity CRP in serum. The visual analog scale (VAS) is a validated patient-reported assessment of functional status and well-being.
Treatment with electrical cymba conchae VNS was evaluated in another open-label, single-arm proof-of-concept trial involving 30 patients with rheumatoid arthritis (RA) who had shown an inadequate response to conventional synthetic disease-modifying antirheumatic drugs (DMARDs) and up to one biologic DMARD. The intervention was well tolerated, and patients experienced clinically meaningful improvements in disease activity, accompanied by reductions in relevant biological markers.
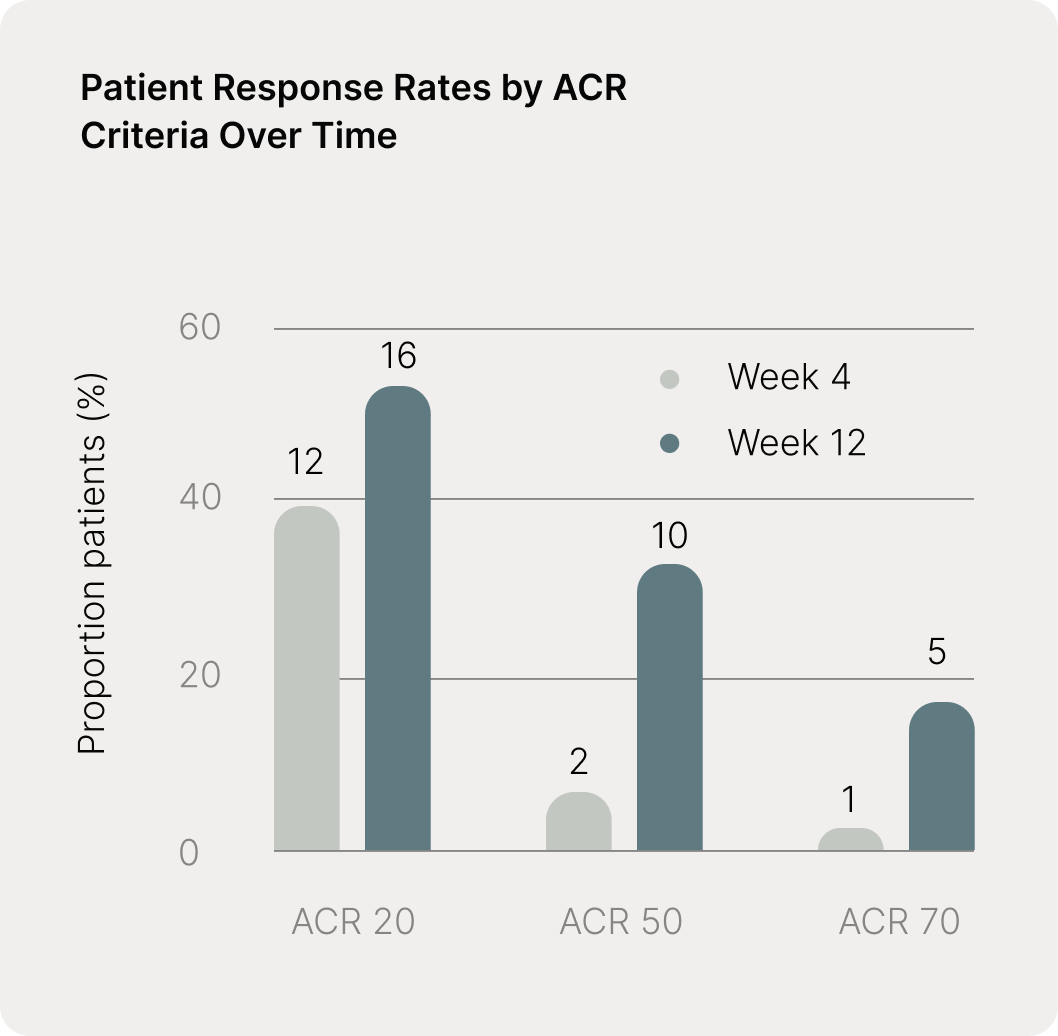
Fig. Proportion of patients (n = 30) achieving ACR20, ACR50, and ACR70 responses at Weeks 4 and 12. ACR20, ACR50, and ACR70 are standardized clinical response criteria commonly used in rheumatoid arthritis (RA) clinical trials and represent ≥20%, ≥50%, and ≥70% improvement, respectively, in core signs and symptoms of RA.
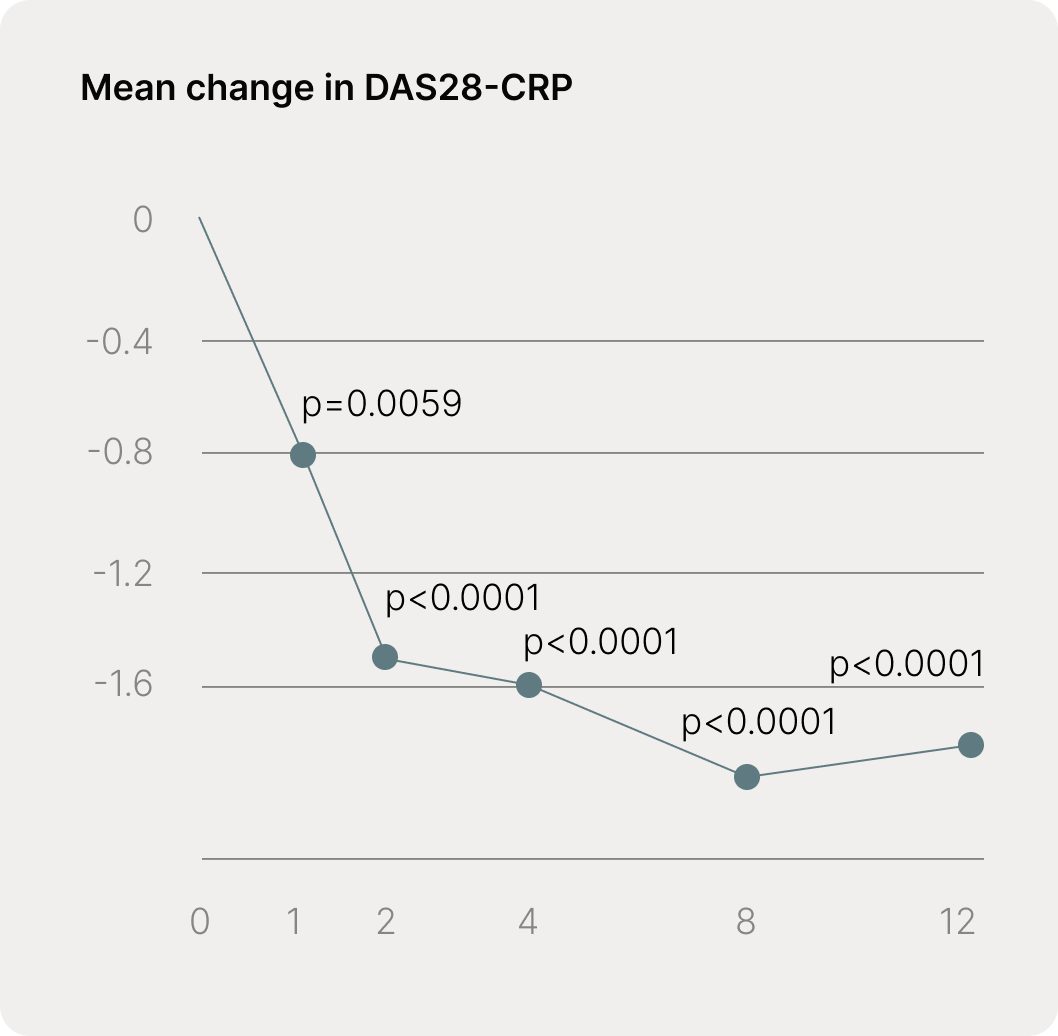
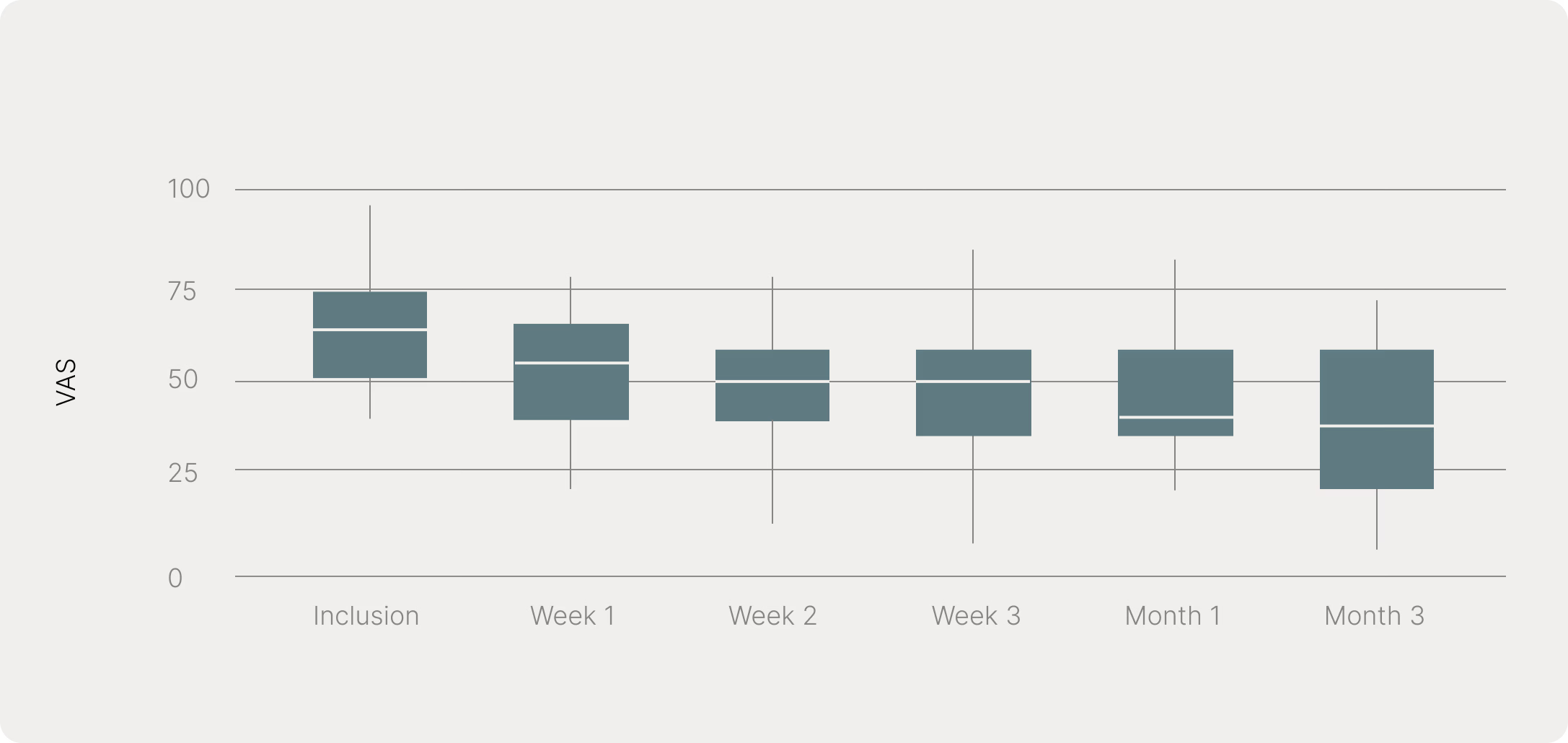
Fig. Mean change in DAS28-CRP for each study visit during the 12-week trial, with p values indicating comparison with baseline, calculated using paired two-tail t-tests. DAS28-CRP is a composite measure of rheumatoid arthritis disease activity based on tender and swollen joint counts (28 joints), C-reactive protein levels, and patient global assessment. CRP reflects presence and severity of inflammation. A DAS28-CRP reduction of 1.4 represents a clinically meaningful improvement and meets established criteria for a moderate or better therapeutic response in RA studies.
1. Adlan, A. M., Lip, G. Y., Paton, J. F., Kitas, G. D. & Fisher, J. P. Autonomic function and rheumatoid arthritis: a systematic review. Seminars in arthritis and rheumatism 44, 283-304, doi:10.1016/j.semarthrit.2014.06.003 (2014).
2. Adlan, A. M. et al. Cardiovascular autonomic regulation, inflammation and pain in rheumatoid arthritis. Autonomic neuroscience : basic & clinical 208, 137-145, doi:10.1016/j.autneu.2017.09.003 (2017).
3. Tesser, J. R. P. et al. Vagus nerve-mediated neuroimmune modulation for rheumatoid arthritis: a pivotal randomized controlled trial. Nat Med, doi:10.1038/s41591-025-04114-7 (2025).
4. Addorisio, M. E. et al. Investigational treatment of rheumatoid arthritis with a vibrotactile device applied to the external ear. Bioelectronic medicine 5, 4, doi:10.1186/s42234-019-0020-4 (2019).
5. Marsal, S. et al. Non-invasive vagus nerve stimulation for rheumatoid arthritis: a proof-of-concept study. The Lancet Rheumatology 3, e262-e269, doi:10.1016/S2665-9913(20)30425-2 (2021).
Chronic Pain

What is chronic pain?
Chronic pain is pain that persists for months or years, often long after an initial injury or inflammation should have healed. It can affect muscles, joints, nerves, or internal organs and is increasingly understood as a condition involving changes in how the nervous system processes and amplifies pain signals.
Common symptoms
People living with chronic pain may experience:
- Persistent or recurring pain lasting months or longer
- Heightened sensitivity to pain, even with mild stimulation
- Fatigue, sleep disturbances, and reduced energy
- Difficulty recovering from physical or emotional stress
- Reduced quality of life and daily function
When the nervous system stays in “alarm mode”
In chronic pain conditions, the nervous system can become overactive and overly sensitive. Instead of calming down after an injury has healed, pain pathways remain switched on—amplifying signals and sustaining discomfort even in the absence of ongoing tissue damage.
This persistent “alarm state” is often linked to ongoing low-grade inflammation and impaired regulation by the autonomic nervous system, which normally helps the body settle, recover, and restore balance.
The role of the vagus nerve
The vagus nerve plays a central role in calming the nervous system and regulating inflammation. It is a key part of the body’s natural pain-control and recovery mechanisms.
Reduced vagal activity has been associated with increased pain sensitivity, fatigue, and diminished ability to recover from stress. By supporting vagus nerve signaling, it may be possible to influence both inflammatory processes and how pain is perceived by the brain.
What clinical research shows
Studies from many sites all over the world demonstrate that non-invasive transcutaneous auricular vagus nerve stimulation (taVNS) is a safe, non-pharmacological approach for managing several forms of chronic pain.
Clinical studies and reviews report that taVNS:
- Reduces pain intensity in chronic and postoperative pain
- Improves fatigue and functional scores in inflammatory conditions such as lupus (SLE)
- Provides clinically meaningful pain relief in conditions like chronic low back pain and irritable bowel syndrome
- Is well tolerated, with no serious adverse events reported in pilot studies
These findings support the role of vagus nerve stimulation as a promising, drug-free approach to pain modulation.
How vagus nerve stimulation may influence pain
How vagus nerve stimulation may influence painPain-sensing nerve cells (nociceptors) release inflammatory molecules such as HMGB1, which contribute to both pain and inflammation. Research shows that activation of the vagus nerve triggers the release of acetylcholine, a neurotransmitter that can suppress HMGB1 release via activation of alpha7 nicotinic acetylcholine receptors and dampen inflammatory pain signaling.
By engaging this built-in regulatory pathway—known as the inflammatory reflex—vagus nerve stimulation may help reduce pain and support recovery at a fundamental biological level.
Key takeaways
- Chronic pain often involves nervous system dysregulation, not just tissue damage
- Persistent inflammation and reduced vagal activity can amplify pain signals
- Clinical research shows that non-invasive ear-based vagus nerve stimulation can reduce pain and fatigue
- taVNS represents a promising, drug-free approach to pain relief grounded in neuroimmunology
Study
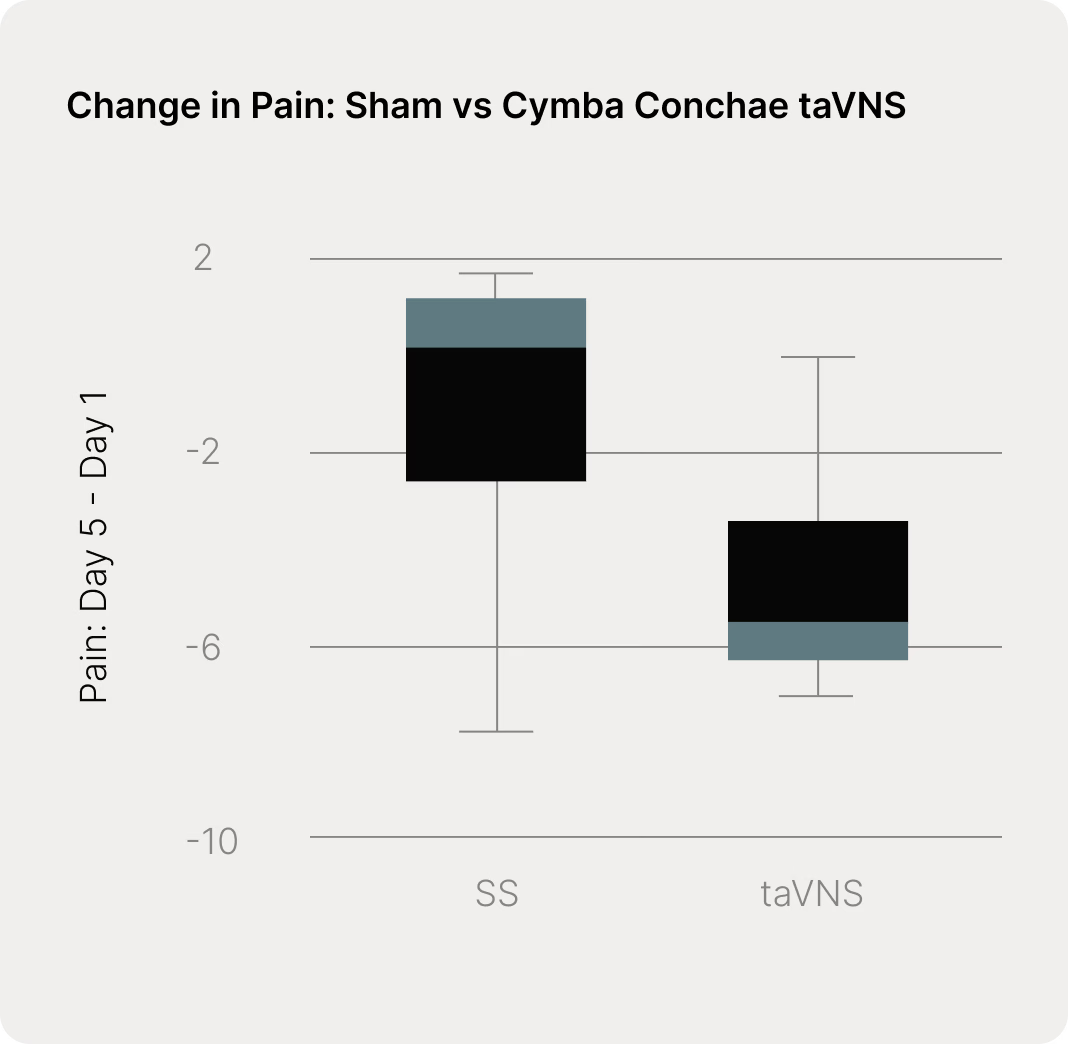
Non-invasive cymba conchae-mediated vagus nerve stimulation alleviates pain in systemic lupus erythematosus (SLE). Patients with chronic systemic inflammatory diseases frequently experience pain and fatigue as major sources of disability. Systemic lupus erythematosus (SLE) is a prototypical example of such a condition. Against this background, it is noteworthy that transcutaneous auricular cymba conchae-mediated vagus nerve stimulation was shown to reduce both pain and fatigue in a randomized, double-blind, sham-controlled pilot trial involving patients with SLE. The cymba conchae area in the ear is exclusively innervated by sensory vagus nerve fibers.
Non-invasive cymba conchae-mediated VNS reduces chronic low back pain. Chronic low back pain (CLBP) represents a major global health burden. A recent proof-of-concept, open-label pilot study with 30 patients evaluated the efficacy, feasibility, and tolerability of transcutaneous cymba conchae vagus nerve stimulation for pain reduction and functional improvement in patients with CLBP. Clinically meaningful pain relief was achieved by 52% of participants, and no serious adverse events were reported.
1. Cohen, S. P., Vase, L. & Hooten, W. M. Chronic pain: an update on burden, best practices, and new advances. Lancet (London, England) 397, 2082-2097, doi:10.1016/s0140-6736(21)00393-7 (2021).
2. Duff, I. T. et al. Clinical Efficacy of Auricular Vagus Nerve Stimulation in the Treatment of Chronic and Acute Pain: A Systematic Review and Meta-analysis. Pain and therapy 13, 1407-1427, doi:10.1007/s40122-024-00657-8 (2024).
3. Chen, J. et al. Transcutaneous Auricular Vagus Nerve Stimulation for Managing Pain: A Scoping Review. Pain management nursing : official journal of the American Society of Pain Management Nurses 26, 33-39, doi:10.1016/j.pmn.2024.11.006 (2025).
4. Aranow, C., Datta-Chadhuri, T., Andersson, U., Chavan, S. & Tracey, K. J. A Randomized, Double-Blind, Sham-Controlled, Clinical Trial of Auricular Vagus Nerve Stimulation for the Treatment of Active Rheumatoid Arthritis: comment on the article by Baker et al. Arthritis & rheumatology (Hoboken, N.J.), doi:10.1002/art.42739 (2023).
5. Tavares-Figueiredo, I. et al. Effect of Transcutaneous Auricular Vagus Nerve Stimulation in Chronic Low Back Pain: A Pilot Study. Journal of clinical medicine 13, doi:10.3390/jcm13247601 (2024).
6. Shi, X. et al. Ameliorating effects and mechanisms of transcutaneous auricular vagal nerve stimulation on abdominal pain and constipation. JCI Insight 6, doi:10.1172/jci.insight.150052 (2021).
7. Yang, H. et al. HMGB1 released from nociceptors mediates inflammation. Proc Natl Acad Sci U S A 118, doi:10.1073/pnas.2102034118 (2021).
8. Yang, H., Andersson, U. & Brines, M. Neurons Are a Primary Driver of Inflammation via Release of HMGB1. Cells 10, doi:10.3390/cells10102791 (2021).
9. Yang, H. et al. Nociceptor α7nAChR activation blunts neuronal HMGB1 release and attenuates inflammation and nociceptive behavior. Mol Med 31, 324, doi:10.1186/s10020-025-01387-z (2025).
10. Fujii, T. & Kawashima, K. An independent non-neuronal cholinergic system in lymphocytes. Japanese journal of pharmacology 85, 11-15, doi:10.1254/jjp.85.11 (2001).
11. Rosas-Ballina, M. et al. Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science 334, 98-101, doi:10.1126/science.1209985 (2011).
Inflammatory Bowel Disease (IBD)

What is inflammatory bowel disease?
Inflammatory bowel disease (IBD) is a group of chronic inflammatory conditions affecting the digestive tract, primarily Crohn’s disease and ulcerative colitis. These conditions are characterized by periods of ongoing intestinal inflammation that can lead to long-term tissue damage and significant impact on daily life.
Common symptoms
People living with IBD may experience:
- Persistent abdominal pain, cramping, and discomfort
- Chronic diarrhea, sometimes with blood
- Urgency and irregular bowel habits
- Fatigue, weight loss, and reduced nutrient absorption
- Periods of flare-ups followed by partial remission
When the gut’s immune system loses balance
The gut contains one of the largest and most complex immune systems in the body. In IBD, immune responses in the intestinal lining remain overactive instead of settling down after their job is done. This leads to persistent inflammation, damage to the gut barrier, and recurring symptoms.
Research shows that this loss of balance is not driven by the immune system alone. It is also closely linked to dysregulation of the nervous system, particularly reduced activity in pathways that normally help calm inflammation.
The role of the vagus nerve
The vagus nerve is the main communication link between the brain and the gut. It plays a central role in regulating digestion, immune responses, and inflammation through the body’s natural anti-inflammatory control mechanisms.
In people with IBD, vagal activity (often referred to as vagal tone) is frequently reduced. This weakened signaling may contribute to ongoing intestinal inflammation and impaired recovery of the gut lining.
What clinical research shows
Clinical research involving scientists from the Feinstein Institutes for Medical Research and Karolinska Institutet indicates that vagus nerve stimulation (VNS) can influence inflammatory activity in the gut.
In a 12-month pilot study of patients with Crohn’s disease, invasive vagus nerve stimulation was well tolerated and associated with reduced inflammatory markers such as CRP and fecal calprotectin, along with improved clinical symptoms. Most participants also showed signs of increased vagal activity.
Emerging evidence for non-invasive ear-based VNS
Early clinical studies suggest that non-invasive transcutaneous auricular vagus nerve stimulation, applied to the cymba conchae—a specific area of the outer ear innervated by vagus nerve fibers—may also benefit patients with IBD.
In a proof-of-concept study involving children and young adults with mild to moderate Crohn’s disease or ulcerative colitis, this approach:
- Improved disease activity scores
- Reduced intestinal inflammatory markers
- Showed a favorable safety profile
Key takeaways
- IBD involves persistent gut inflammation driven by both immune and nervous system dysregulation
- Reduced vagus nerve activity is associated with ongoing intestinal inflammation
- Clinical studies show that vagus nerve stimulation can reduce inflammation and improve symptoms
- Non-invasive ear-based approaches are an active and promising area of clinical research
Study
Significant clinical effects in young patients with IBD using non-invasive VNS. A small proof-of-concept study in children and young adults with mild to moderate Crohn’s disease or ulcerative colitis showed that non-invasive transcutaneous auricular VNS applied in the cymba concha region improved symptoms and reduced inflammatory markers including fecal calprotectin. There were no safety issues.
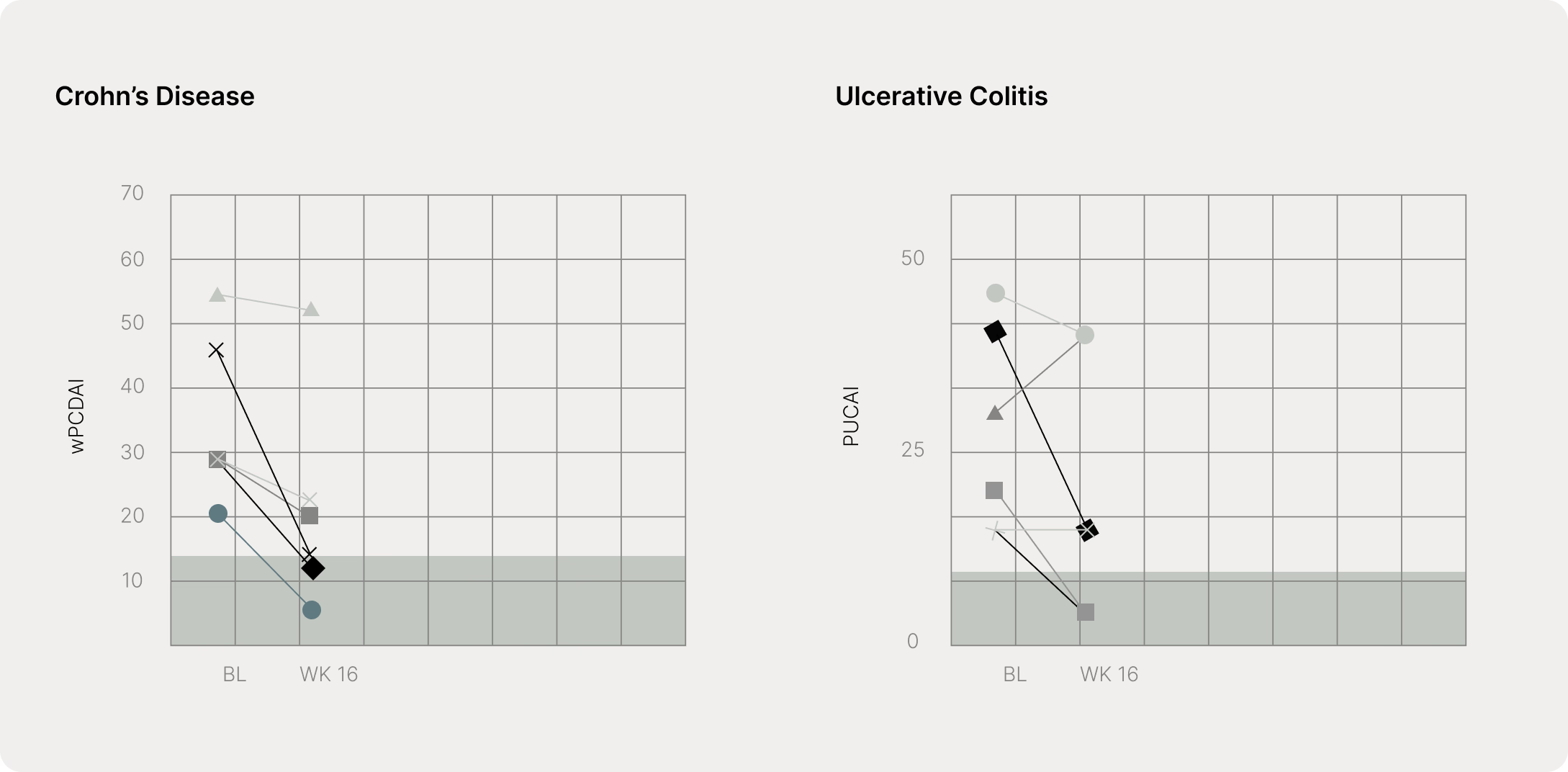
Fig. Change in disease activity index from baseline (BL) to week (WK) 16. a wPCDAI = weighted pediatric Crohn’s disease activity index. b PUCAI = pediatric ulcerative colitis activity index. Shaded areas represent remission zones.
1. Kim, K. N., Yao, Y. & Ju, S. Y. Heart rate variability and inflammatory bowel disease in humans: A systematic review and meta-analysis. Medicine 99, e23430, doi:10.1097/md.0000000000023430 (2020).
2. Sinniger, V. et al. A 12-month pilot study outcomes of vagus nerve stimulation in Crohn's disease. Neurogastroenterology and motility : the official journal of the European Gastrointestinal Motility Society 32, e13911, doi:10.1111/nmo.13911 (2020).
3. Sahn, B., Pascuma, K., Kohn, N., Tracey, K. J. & Markowitz, J. F. Transcutaneous auricular vagus nerve stimulation attenuates inflammatory bowel disease in children: a proof-of-concept clinical trial. Bioelectronic medicine 9, 23, doi:10.1186/s42234-023-
Long COVID

What is Long COVID?
Long COVID refers to symptoms that persist for months or years after an acute COVID-19 infection. These symptoms can affect multiple body systems and often fluctuate over time, making daily activities, work, and recovery difficult for many people.
Common symptoms
People living with Long COVID may experience:
- Persistent fatigue and reduced energy levels
- Brain fog, concentration difficulties, and cognitive decline
- Worsening of symptoms after physical or mental exertion (post-exertional symptom exacerbation)
- Shortness of breath and reduced exercise tolerance
- Heart rate instability and other signs of autonomic dysfunction
When recovery after infection is disrupted
After a viral infection, the body normally resolves inflammation and returns to a balanced state. In Long COVID, this recovery process appears to be impaired. Research indicates that inflammation remains active for too long, either throughout the body or within specific tissues.
This ongoing, poorly regulated inflammation can disrupt normal physiological stress responses and contribute to persistent symptoms such as fatigue, cognitive impairment, and reduced tolerance to exertion.
The role of the vagus nerve
The vagus nerve plays a central role in regulating inflammation and restoring balance after immune activation through a mechanism known as the inflammatory reflex.
Emerging evidence shows that SARS-CoV-2 can infect and inflame parts of the vagus nerve and connected brain regions. Such involvement may weaken the body’s natural ability to control inflammation, either temporarily or over the long term, potentially contributing to the symptoms seen in Long COVID.
What research shows
Work at many research centers around the world including the Karolinska Institutet and The Feinstein Institutes for Medical Research has identified signs of vagus nerve dysfunction in individuals with Long COVID. Early pilot studies and case reports suggest that non-invasive vagus nerve stimulation, including stimulation applied to the cymba conchae region of the ear, may support autonomic regulation and recovery.
While these initial findings are encouraging, current evidence is based on small studies with varying designs. Larger, well-controlled clinical trials are ongoing to more clearly evaluate the potential role of vagus nerve stimulation in Long COVID.
Key takeaways
- Long COVID is associated with persistent symptoms and impaired recovery after SARS-CoV-2 infection
- Dysregulated inflammation and autonomic nervous system imbalance may play a central role
- The vagus nerve is essential for controlling inflammation and supporting recovery
- Early research suggests neuromodulation may help restore balance, but further studies are needed
1. Woo, M. S. et al. Vagus nerve inflammation contributes to dysautonomia in COVID-19. Acta neuropathologica 146, 387-394, doi:10.1007/s00401-023-02612-x (2023).
2. Andersson, U. & Tracey, K. J. Vagus nerve SARS-CoV-2 infection and inflammatory reflex dysfunction: Is there a causal relationship? J Intern Med 295, 91-102, doi:10.1111/joim.13746 (2024).
3. Pfoser-Poschacher, V. et al. Feasibility and acceptance of transdermal auricular vagus nerve stimulation using a TENS device in females suffering from long COVID fatigue. Wiener klinische Wochenschrift 137, 635-641, doi:10.1007/s00508-025-02501-1 (2025).
4. Khan, M. W. Z. et al. Vagal nerve stimulation for the management of long COVID symptoms. Infectious medicine 3, 100149, doi:10.1016/j.imj.2024.100149 (2024).
5. Kranck, G., Ståhlberg, M., Andersson, U., Lundin, J. & Fedorowski, A. Monitoring of cardiorespiratory vagal desynchrony using novel biomarkers derived from smartwatch electrocardiograms in a patient recovering from long COVID: case report. European heart journal. Case reports 9, ytaf425, doi:10.1093/ehjcr/ytaf425 (2025).
The information presented is based on published scientific research and is intended for educational purposes only. Vagus nerve stimulation is an active area of clinical research and is not yet approved for all conditions described. Always consult a qualified healthcare professional regarding diagnosis or treatment decisions.