HRV can tell us a lot about general well-being—but much less about whether vagus nerve stimulation is actually activating the body’s immune-regulating pathways.
What HRV really measures
A window into autonomic balance—not immune control
Heart rate variability (HRV) refers to the natural variation in time between heartbeats. Even at a steady heart rate, the intervals between beats are never perfectly equal. This variability reflects how the autonomic nervous system is functioning.
HRV is influenced by two systems working together:
- The sympathetic nervous system, which speeds the heart up (stress, activity)
- The parasympathetic nervous system, largely mediated by the vagus nerve, which slows the heart down (rest, recovery)
A correct interpretation of a high HRV value is that it reflects the balance between the parasympathetic and sympathetic systems rather than conveying an assessment of the vagus system’s isolated influence on HRV. In the setting of chronic inflammatory diseases, the vagal activity is blunted as the vagus is not optimally activated by endogenous systems. Therefore, direct vagal stimulation restores toward optimal, shifting the parasympathetic/ sympathetic balance back towards parasympathetic.
Why this matters for vagus nerve stimulation
Each of the two vagus nerves contains around 100,000 nerve fibers, and these fibers serve very different functions. About 80% carry sensory information from the body to the brain, while only around 20% carry motor signals from the brain to organs.
The inflammatory reflex—the brain’s built-in brake on excessive inflammation—is mediated by specific vagal pathways, not by all vagus-controlled functions. As a result, changes in heart rhythm do not reliably indicate whether the immune-regulating part of the vagus nerve has been activated.
In clinical research, activation of the inflammatory reflex is confirmed through blood-based biomarkers, such as reductions in inflammatory molecules. Unfortunately, no at-home test can currently measure this directly.
Why HRV still matters for wellness
Useful—but for a different question
For outcomes such as stress reduction, sleep quality, or general recovery, HRV can be a helpful indicator. Whether HRV improves because vagal activity increases or because sympathetic activity decreases, both mechanisms are often associated with better well-being.
This is why HRV is widely used—and useful—in wellness contexts.
But wellness is not the same as immune regulation.
Why HRV can mislead in inflammation and disease
The cholinergic anti-inflammatory pathway—the core mechanism of the inflammatory reflex—is strictly mediated by the vagus nerve. A reduction in sympathetic activity alone does not activate this pathway.
Because HRV combines signals from both systems, it cannot distinguish:
- true vagal activation from
- reduced sympathetic drive
As a result, HRV may improve without meaningful engagement of the inflammatory reflex. This makes HRV an unreliable biomarker for evaluating whether vagus nerve stimulation is modulating immune function.
A better indicator: heart–breathing synchronization
When timing matters more than variability
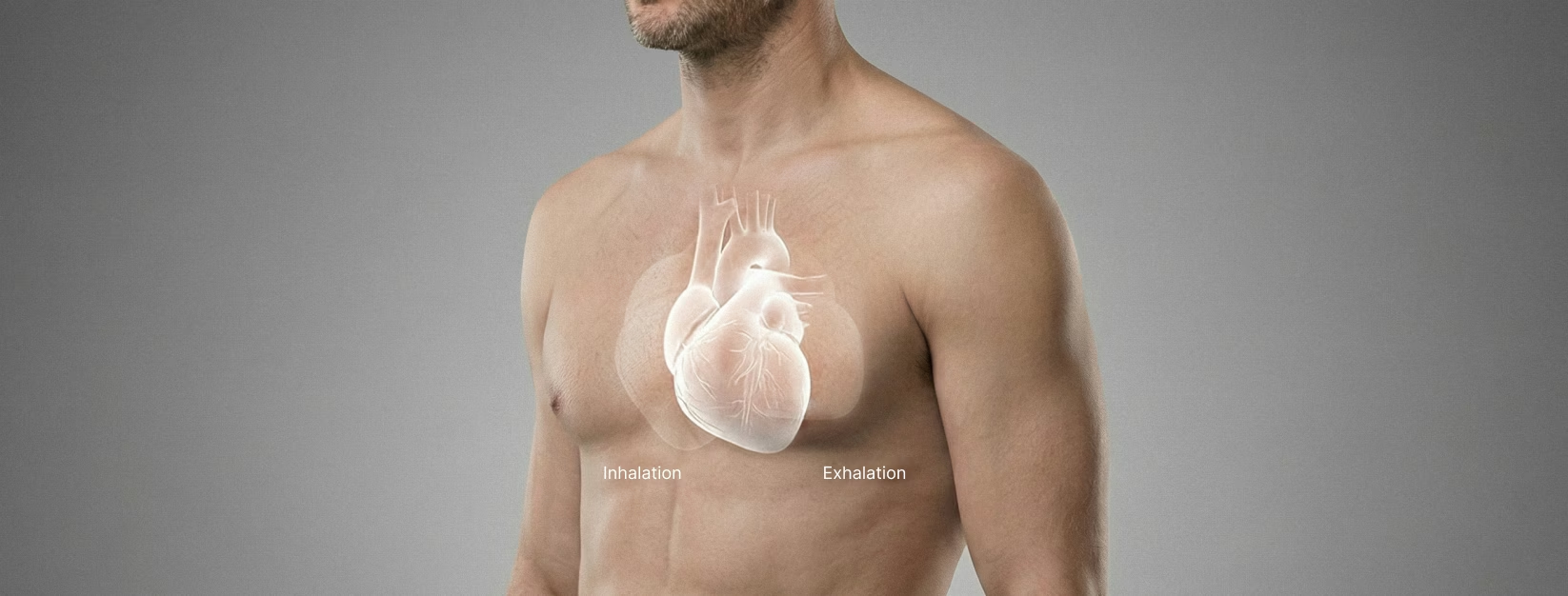
There is a more vagus-specific signal—if heart rate and breathing are measured at the same time.
In a healthy system:
- Heart rate increases during inhalation
- Heart rate decreases during exhalation
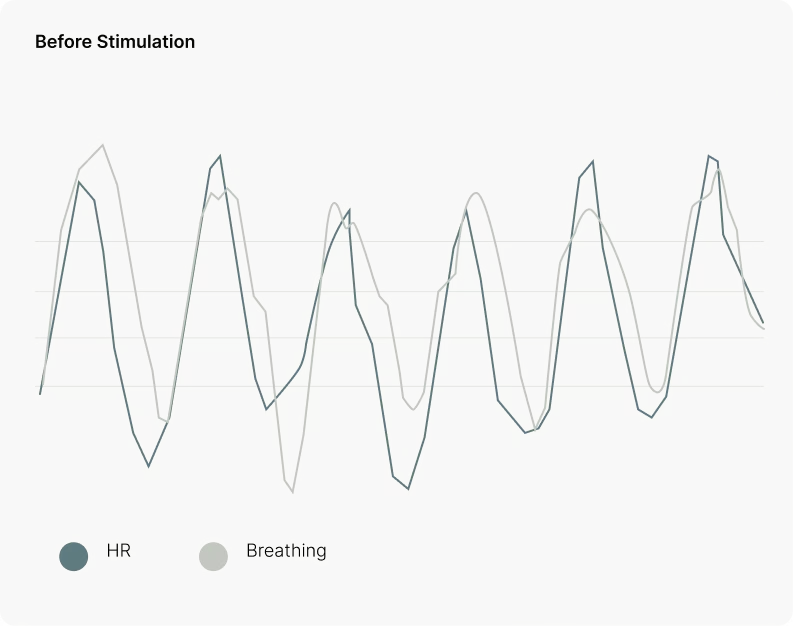
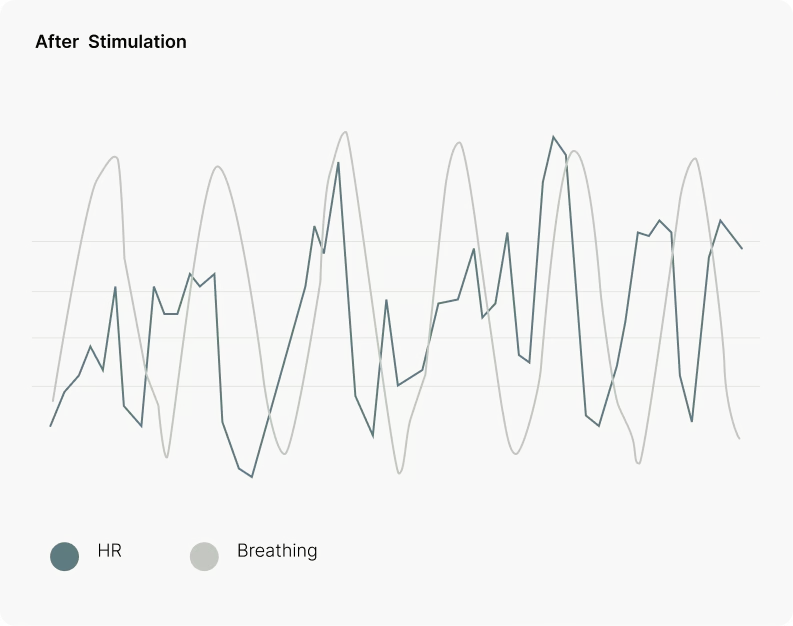
This phenomenon, called respiratory sinus arrhythmia, is controlled almost entirely by the vagus nerve. When heart rhythm and breathing are well synchronized, oxygen delivery is optimized and vagal regulation is strong.
Importantly, a person may have a normal or high HRV but poor heart–breathing synchronization, offering little physiological benefit.
Can smartwatches measure this?
Surprisingly, yes. But how can a wrist-worn watch measure breathing using smart biosensing? During slow, controlled inhalation and exhalation, the diaphragm moves and subtly rotates the heart because the pericardium is connected to the diaphragm. These tiny movements alter the electrical signals detected by the ECG sensor of the watch. Using advanced algorithms, the watch converts these signal patterns into a clear, continuous representation of the breathing pattern. This allows simultaneous tracking of heart rhythm and respiration—making it possible to assess cardio-respiratory synchronization, a more direct marker of vagal function.
Early research suggests this approach may provide better insight into vagal regulation than HRV alone, particularly in recovery from conditions such as Long COVID.1