The brain can help restore immune balance through signals carried in the vagus nerve. When this regulatory system is not functioning properly, inflammation may stay switched on—what we often call chronic inflammation. Therapies based on electrical vagus nerve stimulation are therefore being explored as a way to support the body’s own ability to calm dysregulated inflammation and, in some cases, reduce symptoms across multiple organ systems.1
A therapy with decades of clinical use
From epilepsy and depression to inflammation research
Implanted vagus nerve stimulation has been used for decades in neurology and psychiatry, particularly for epilepsy and treatment-resistant depression, with a well-established safety profile and few severe adverse events.2,3
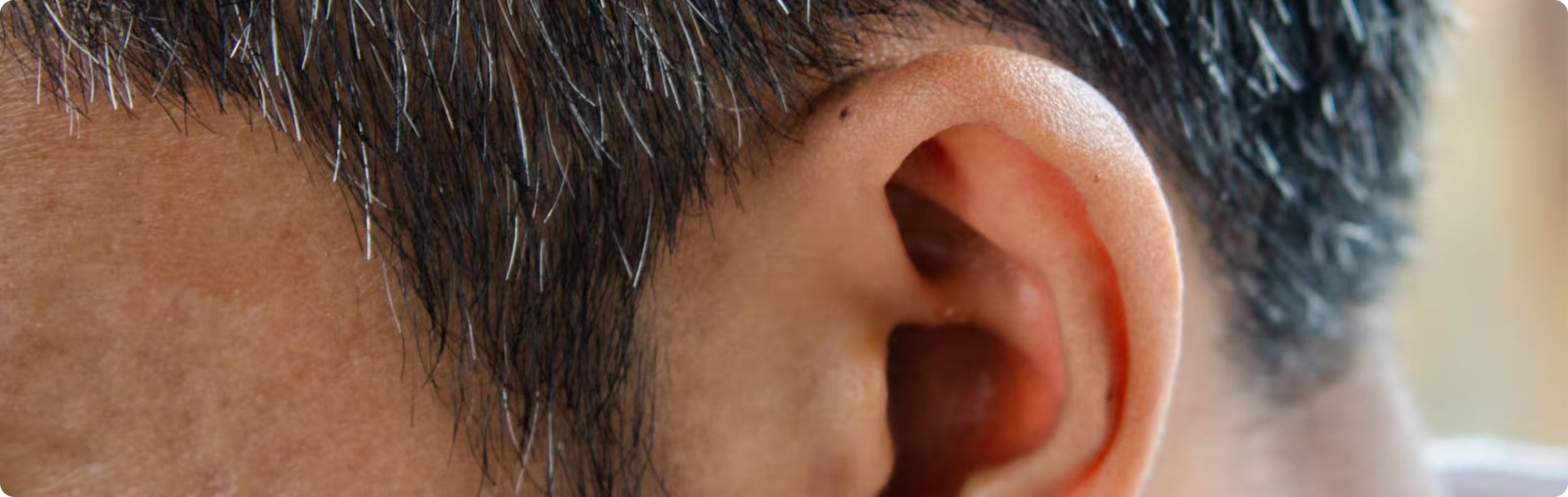
More recently, non-invasive methods have been developed. In transcutaneous auricular vagus nerve stimulation (taVNS), small electrodes are placed on specific regions of the outer ear that are innervated by sensory fibers of the vagus nerve. This external approach has been used increasingly over the past decade in the same clinical areas where implanted VNS has long been applied.4-7
First clinical breakthrough in inflammatory disease
A rheumatoid arthritis case that opened the door
The first successful report of treating a chronic inflammatory disease using implanted VNS was published in 2012, describing a patient with therapy-resistant rheumatoid arthritis (RA) who improved enough to return to normal life.8
Since then, multiple studies have supported the idea that invasive VNS can influence inflammation and symptoms in conditions such as RA and inflammatory bowel disease, and also in inflammation-linked neurological conditions.9-15
Stronger evidence: a pivotal RA trial and FDA approval
A milestone for bioelectronic medicine
A major step forward came with a pivotal randomized, double-blind, placebo-controlled trial in RA (242 patients), published in December 2025.9 The study reported sustained clinical benefits, high patient satisfaction, and low rates of adverse events through 12 months of follow-up in patients with active RA, that had been refractory to conventional therapies.
Importantly, the trial’s results supported FDA approval (July 2025) of an implanted VNS device for treating adults with moderately to severely active RA—an historic milestone as the first regulatory endorsement of vagus nerve stimulation for an inflammatory disease.9
Long-term follow-up data from earlier RA cohorts have also reported sustained efficacy and favorable safety over multiple years.11
The rise of non-invasive ear-based VNS (taVNS)
Promising results with mild, temporary side effects
Kevin Tracey’s research group was also among the first to show that non-invasive ear-based VNS could be used with encouraging outcomes in rheumatoid arthritis.16
Subsequent pilot studies have reported promising findings across additional conditions, including RA, inflammatory bowel disease, systemic lupus erythematosus (SLE), pediatric kidney inflammation, and several chronic pain conditions.17-20
Across systematic reviews and pooled safety analyses, taVNS is generally reported as safe and well tolerated, with most side effects being mild and temporary—most commonly skin irritation or local discomfort at the stimulation site.4-7
Well-controlled trials are now ongoing worldwide to better define the therapeutic potential of non-invasive VNS for chronic inflammation and chronic pain.