For a long time, inflammation was seen as something the immune system handled on its own. That changed when Dr Kevin Tracey and his team at The Feinstein Institutes for Medical Research uncovered a surprising truth: the brain doesn’t just respond to inflammation — it can actively control it through signals carried in the vagus nerve.1-4
This discovery revealed the inflammatory reflex: a brain-controlled neural circuit that senses rising inflammation and sends “calming” signals back to the immune system to prevent inflammation from becoming excessive or chronic.1 It also helped create a new medical field—bioelectronic medicine—where the goal is to use targeted nerve stimulation to influence specific biological pathways, rather than relying only on drugs.1-4
Why this matters
The body’s built-in brake on runaway inflammation
Inflammation is essential for healing, but too much of it can be damaging. The inflammatory reflex acts like an internal braking system—helping keep immune activity within healthy limits.1,3 When that brake is impaired, inflammation may stay switched on, increasing the risk of tissue damage and contributing to chronic disease.
This concept became even more relevant during COVID-19. Autopsy studies have shown inflammation in the vagus nerve and connected brainstem regions in people who died during severe infection.5 Researchers have raised the possibility that vagus involvement could impair the body’s normal inflammatory control mechanisms, potentially contributing to extreme immune reactions such as cytokine storms.6
The “accident” that started a breakthrough
A drug in the brain changed inflammation in the whole body
One of the turning points was an unexpected laboratory finding in the 1990s. Tracey’s team studied an experimental compound known to suppress the inflammatory molecule TNF. When researchers injected this compound into the brains of animals, it blocked TNF in the brain—exactly as expected.2
But something surprising happened: TNF was also suppressed in peripheral organs like the spleen and liver, even though the compound could not cross the blood–brain barrier to reach those organs. That meant the brain must be sending a signal to the immune system — fast.
Tracey’s team tested which “wire” carried the signal. When they surgically cut the left vagus nerve, the anti-inflammatory effect disappeared. When they stimulated the vagus nerve electrically, they could reproduce the effect without the drug.2
That was the key insight: electrical signals in the vagus nerve can regulate inflammation throughout the body.
How the inflammatory reflex works
From immune sensing → brain decision → immune calming
Reflexes are how the brain automatically keeps your body stable—heart rate, breathing, digestion. Tracey’s insight was that the immune system should have reflex control too.1,3 The inflammatory reflex has two main “arms”:
1) The sensory arm (signals going up to the brain)
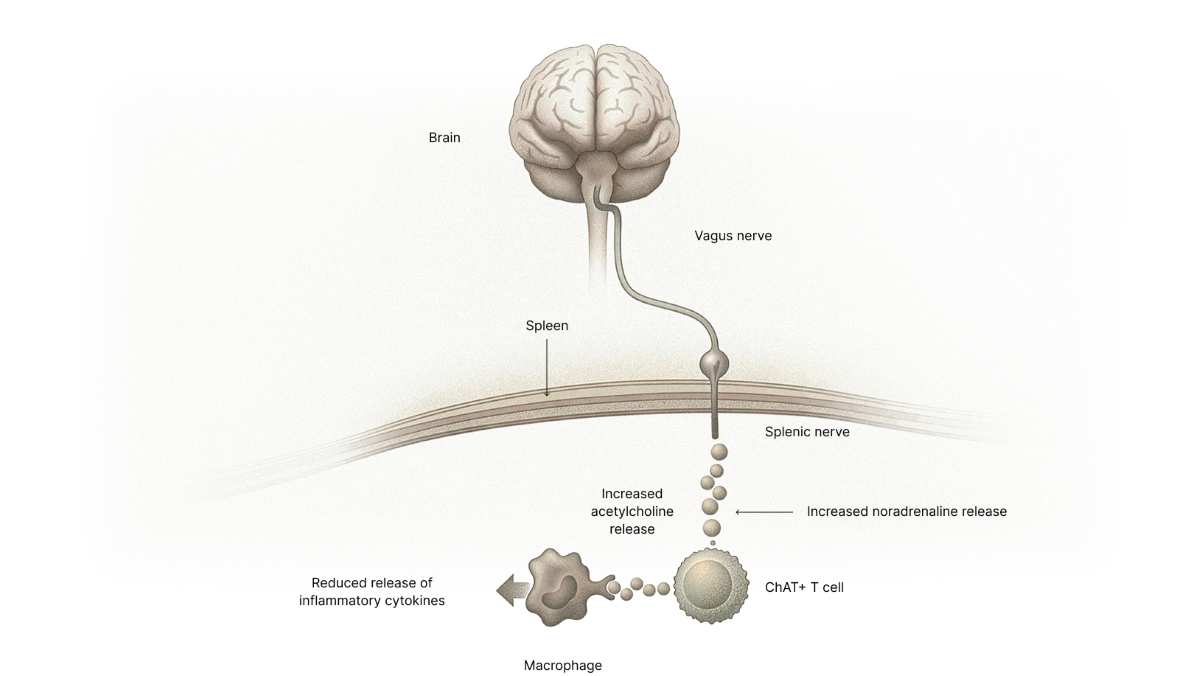
Your body continuously monitors what’s happening inside—especially when tissues are stressed or inflamed. Sensory fibers in the vagus nerve relay these inflammatory signals to the brainstem,7 particularly a hub called the nucleus tractus solitarius (NTS).
Research has shown that sensory vagus activity changes in response to specific cytokines and inflammatory signals — and that these patterns can be decoded.8-10 Scientists have also identified a role for specific sensory fibers (including TRPA1-expressing nociceptors) in triggering reflex anti-inflammatory responses.10
2) The efferent arm (signals going back down to the immune system)
Signals traveling through the vagus nerve reach key relay points in the abdomen and connect into pathways that influence the splenic nerve.3,4 In the spleen, nerve signaling triggers the release of noradrenaline, which activates specialized T cells (ChAT+) that can produce acetylcholine and move to inflammatory sites, also to areas not innervated by the vagus system.11
Acetylcholine then binds to receptors (notably α7 nicotinic acetylcholine receptors) on immune cells such as macrophages, helping suppress inflammatory pathways and reduce cytokines like TNF.1-4
This cholinergic signaling has also been linked to reduced release of HMGB1, an inflammatory molecule involved in severe inflammation—and even pain signaling through nociceptors.12-14
From discovery to human studies
Turning biology into real-world therapy
The inflammatory reflex is not only a laboratory concept. The pathway has been demonstrated in humans using vagus nerve stimulation (VNS), including clinical research in rheumatoid arthritis and other inflammatory conditions.15,16
Researchers have also explored non-invasive, ear-based stimulation (transcutaneous auricular VNS) that targets the cymba conchae, one of the rare skin areas with vagus sensory innervation.17 Early clinical studies have reported encouraging outcomes in inflammatory and pain-related conditions, including IBD, lupus, and rheumatoid arthritis with ongoing research continuing to refine best protocols.18-20