By studying the effects of auricular stimulation in animal models, researchers have been able to explore how vagus nerve signaling influences inflammation, pain, and the neural pathways connecting the brain and immune system.
These studies provide early insights into how stimulating the vagus nerve through the outer ear may engage the body’s natural regulatory mechanisms.
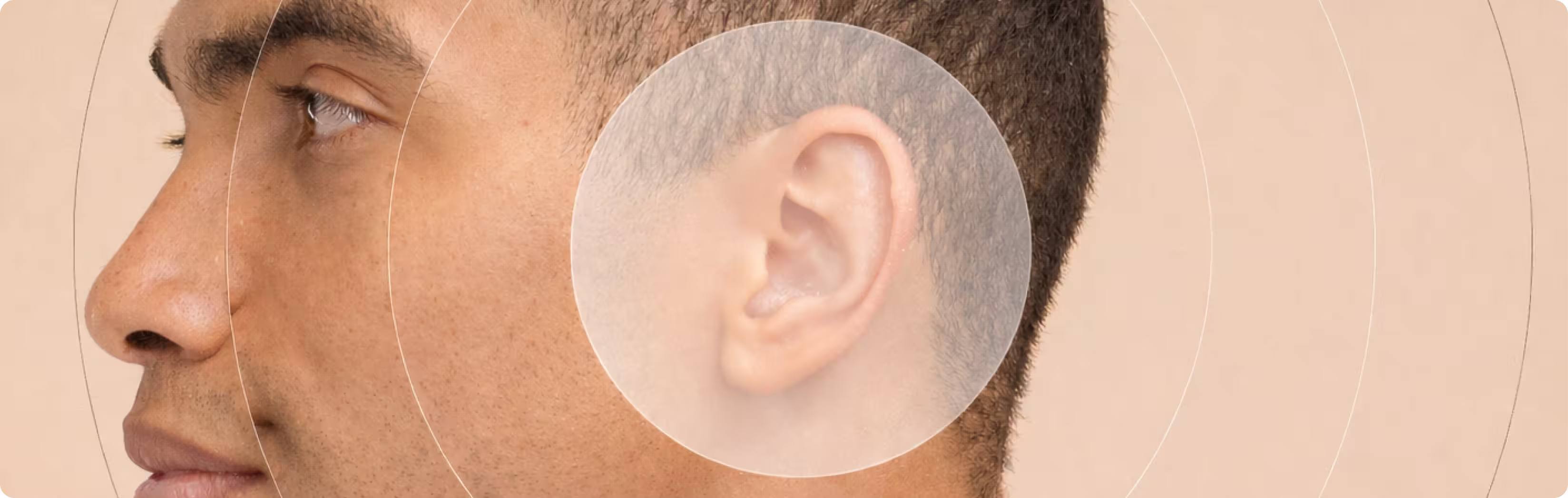
A unique access point in the outer ear
The outer ear holds a rare connection to the vagus nerve.
It is the only location on the skin where branches of the vagus nerve reach the surface. This makes it possible to stimulate the nerve non-invasively through specific areas of the ear.
Because this anatomical feature exists across many mammals, it has allowed researchers to study vagus nerve stimulation in animal models before translating these findings to humans.
Evolutionary research provides a possible explanation. Parts of the mammalian outer ear developed from structures related to fish gill arches. Since the vagus nerve plays an important role in controlling gill function in fish, some of these nerve connections appear to have been preserved as these structures evolved into the mammalian ear.
Today, this small nerve branch in the ear provides a natural entry point for external vagus nerve stimulation.
What animal studies show about inflammation
Research in animal models indicates that stimulating the ear can influence inflammatory activity throughout the body.
In several studies, auricular vagus nerve stimulation has been associated with reduced levels of inflammatory signaling molecules, including:

These molecules are important regulators of immune responses during inflammation.
Importantly, when researchers experimentally block vagus nerve signaling, these effects disappear. This indicates that the responses observed in these studies are mediated through vagus nerve pathways.
Together, these findings support the role of vagus nerve signaling in the body’s mechanisms for regulating inflammation.
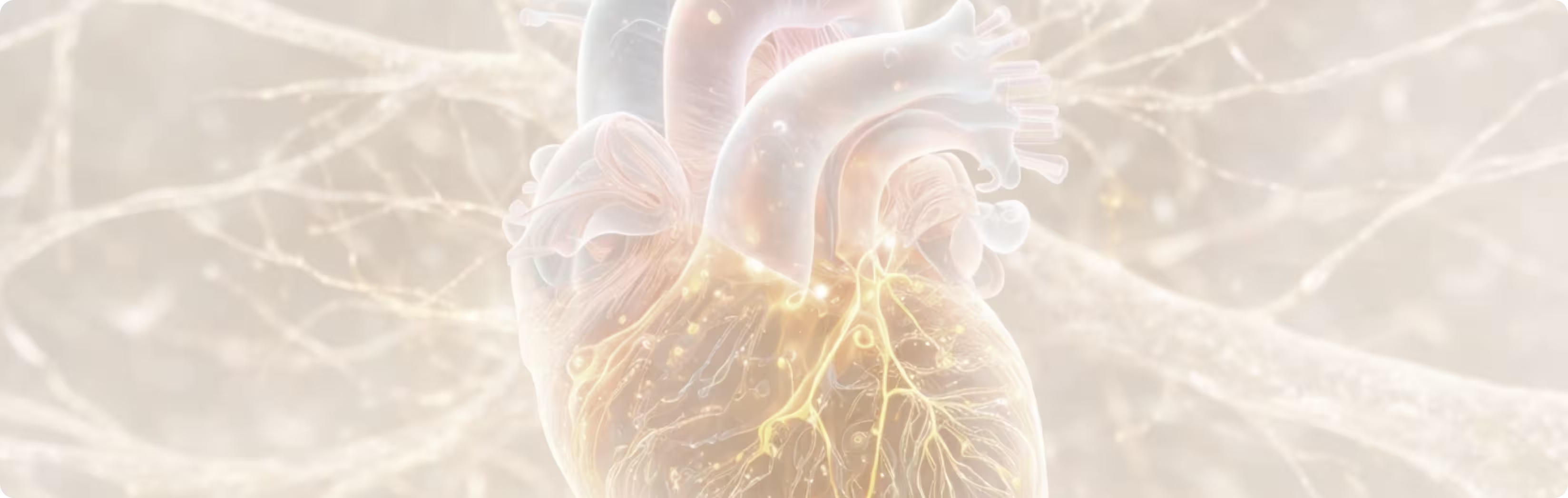
How signals travel from the ear to the brain
Although stimulation is applied to the ear, the signals travel through neural pathways that extend to the brain.
Signals from the auricular branch of the vagus nerve reach the brainstem, where they activate networks involved in autonomic regulation. These neural circuits help coordinate functions such as immune activity, cardiovascular regulation, and recovery processes.
Animal studies have also helped researchers explore how different stimulation settings—such as frequency and intensity—affect these neural circuits. This work has helped guide the development of stimulation protocols used in current research.
Links between inflammation and pain
Inflammation and chronic pain are closely connected through interactions between the nervous and immune systems.
In animal models, auricular vagus nerve stimulation has been associated with reductions in pain-related behaviors alongside decreases in markers of neuroinflammation.
Researchers believe these effects may involve several mechanisms, including:
- modulation of immune activity in the brain and spinal cord
- reduced activation of inflammatory cells such as microglia
- activation of the body’s natural pain-regulating neural pathways
These findings highlight the close relationship between neural signaling, inflammation, and pain.
Safety insights from preclinical research
Animal studies have also provided valuable information about safety.
When stimulation is delivered within commonly used research parameters, auricular vagus nerve stimulation has generally been well tolerated in animal models.
Because the vagus nerve plays a role in regulating heart rate and blood pressure, very strong stimulation can influence cardiovascular reflexes. Reassuringly, the current required to engage vagal circuits regulating inflammation is far lower than that required for cardiac control. These findings help researchers define safe stimulation ranges and highlight the importance of carefully controlled stimulation parameters.